Posted on November 26, 2025
Keywords: Medical Laser Marking Machine, UDI Laser Marking, UV Laser Marking, Fiber Laser Marking, ISO 13485
The medical device industry operates under stringent regulatory requirements, most notably the global Unique Device Identification (UDI) system mandated by the FDA and EU MDR. Choosing the right laser marking machine is not merely a technical decision; it is a critical compliance and quality assurance choice. The mark must be permanent, legible, and non-damaging to the device material, especially after sterilization and repeated use. This guide provides a comprehensive framework for selecting a medical laser marking machine that ensures both UDI laser marking compliance and optimal production efficiency.
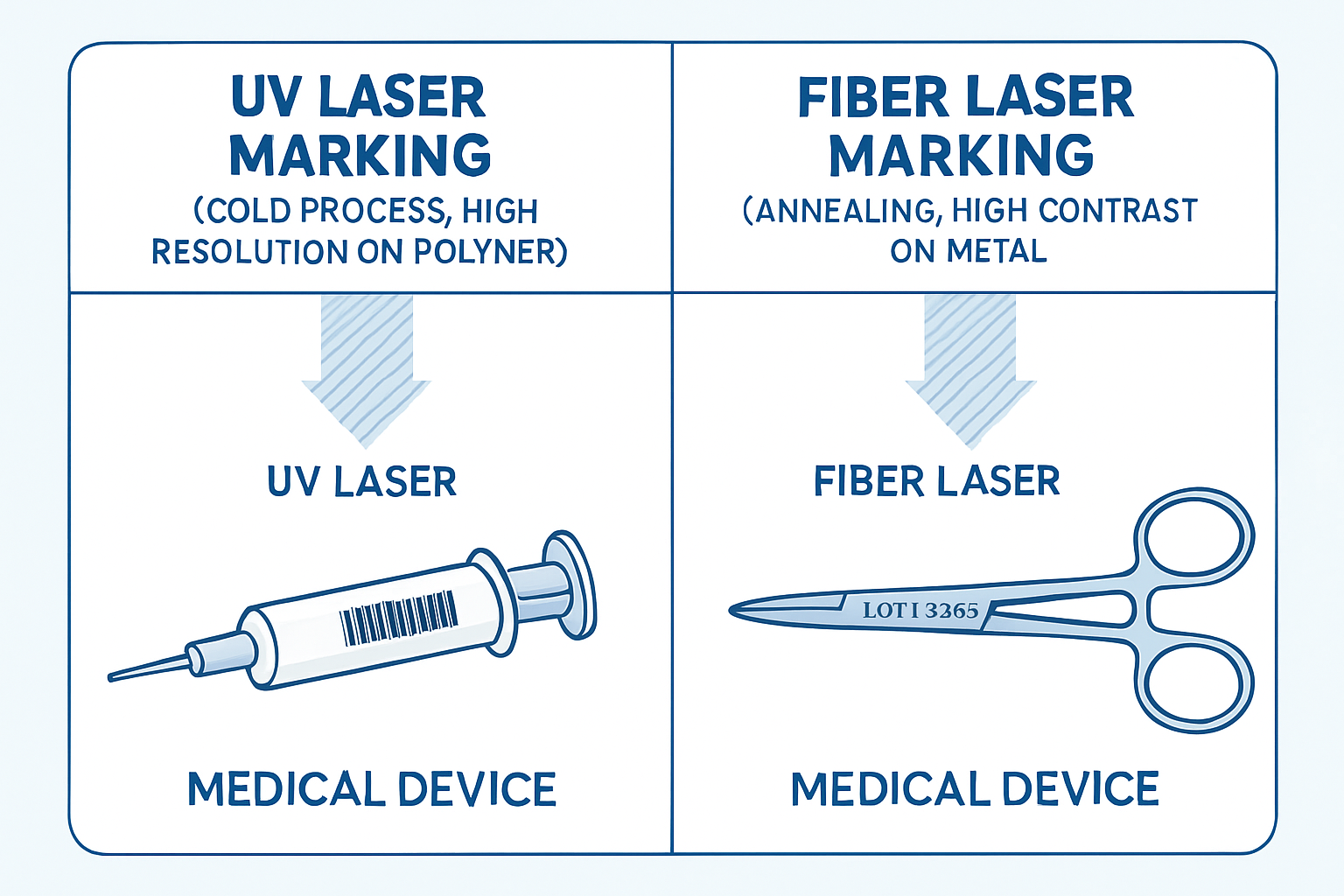
The first step in selection is matching the laser wavelength to the material of your medical device. Medical devices utilize a wide range of materials, each reacting differently to laser energy.
For materials like stainless steel, titanium, and anodized aluminum, the Fiber Laser Marking machine (typically 1064nm wavelength) is the industry standard.
Heat-sensitive materials like PEEK, ABS, and various plastics require a gentler approach to prevent thermal damage, micro-cracks, or material degradation. This is where the UV Laser Marking machine (typically 355nm wavelength) excels.
Compliance extends beyond the physical mark itself. Your laser system must be part of a validated process.
The UDI mark must be readable by both humans and machines (e.g., Data Matrix codes). Key considerations include:
Medical device manufacturers must validate their processes according to ISO 13485 standards. Your laser marking supplier should be able to provide comprehensive support for:
Beyond material compatibility, several technical specifications impact performance and quality:
| Parameter | Importance | Why It Matters |
|---|---|---|
| Beam Quality ($M^2$) | High | A lower $M^2$ value (closer to 1) indicates a higher quality beam that can be focused to a smaller spot size, crucial for ultra-fine marking on small devices. |
| Galvo Speed | Moderate | Determines the marking speed (characters per second). High speed is necessary for high-volume production lines. |
| Spot Size | High | Directly impacts the resolution and minimum feature size. Essential for marking tiny components like stents or small surgical tools. |
| Integrated Vision System | Critical | A built-in camera and software to verify the mark's quality (e.g., contrast, legibility, data content) immediately after marking, ensuring 100% quality control. |
A modern medical laser marking system must be designed for seamless integration into an automated production line.
Selecting a medical laser marking machine is a long-term investment in quality and compliance. By prioritizing material compatibility (UV for plastics, Fiber for metals), demanding robust UDI validation support (ISO 13485), and evaluating key technical parameters, you can ensure your marking process is both compliant and highly efficient. Partner with a supplier who understands the unique demands of the medical device lifecycle.
Figure 1: Comparison of Laser Marking Technologies for Medical Devices
Explore more insights to inform your business decisions