لماذا يُعدّ وضع العلامات بالليزر ضروريًّا للامتثال لمتطلبات المعرفة الفريدة للأجهزة (UDI) الخاصة بالمنظار الداخلي وتحقيق إمكانية التتبّع
متطلبات إدارة الأغذية والأدوية الأمريكية (FDA) المتعلقة بالمعرفة الفريدة للأجهزة (UDI) الخاصة بالمناظير المرنة والصلبة: كيف يحقّق وضع العلامات بالليزر متطلبات الدوام وسهولة القراءة
تتطلب إدارة الأغذية والعقاقير الأمريكية (FDA) تسميات تعريف الأجهزة الفريدة (UDI) على المناظير الطبية لأغراض التتبع ولضمان سلامة المرضى أثناء الإجراءات. ووفقاً للوائح، يجب أن تبقى هذه العلامات ثابتة بشكل دائم، وأن تكون واضحة للعين، وأن تتحمل عمليات التنظيف المتكررة والتعامل الشديد في المستشفيات. وتُعد طريقة الوسم بالليزر فعّالة جداً في هذا السياق، لأنها تُغيّر المواد تحت سطح القطعة بدل أن تكتفي بخدش السطح أو طلائه. وهذا يعني أن العلامات لن تتآكل حتى بعد التعرّض للمواد الكيميائية القاسية أو الخضوع لمئات دورات التعقيم في أجهزة التعقيم الحرارية (الأوتوكلاف) الساخنة. أما الملصقات العادية أو الطلاء فلا تثبت مع مرور الوقت. وأظهرت الاختبارات أن علامات UDI المحفورة بالليزر تظل مقروءة بعد أكثر من ٥٠٠ دورة تعقيم عند درجة حرارة تبلغ نحو ١٣٤ درجة مئوية، مما يتوافق مع المعايير المنصوص عليها في لائحة إدارة الأغذية والعقاقير الأمريكية ٢١ CFR الجزء ٨٠١. ومن المزايا الكبرى الأخرى أن الليزر يترك الأسطح ناعمة وخالية من الشقوق الدقيقة التي قد تختبئ فيها البكتيريا، ما يجعلها أكثر أماناً للمرضى. كما أنه يعمل بكفاءة عالية على الأشكال المعقدة سواء كان المنظار صلباً أم مرنًا.
المتانة المقارنة: الوسم بالليزر مقابل الطباعة النافثة للحبر، والنقش، والتجويه في البيئات السريرية عالية الإجهاد
عندما يتعلق الأمر بالتطبيقات السريرية الفعلية، فإن الوسم بالليزر يتفوق على الطرق التقليدية لأنه يُحدث اندماجًا فعليًّا للمواد على المستوى الجزيئي، بدلًا من مجرد وضع مواد سطحية أو إحداث تلفٍ ماديٍّ في الأسطح. أما المشكلات المرتبطة بالطرق الأخرى فهي جوهريةٌ جدًّا. فعلى سبيل المثال، تميل الملصقات النافثة للحبر إلى التفكك بعد نحو ٢٠ إلى ٣٠ دورة تعقيم، إذ تذيبها المواد الكيميائية المستخدمة في هذه العمليات. كما أن النقش الميكانيكي يُحدث شقوقًا دقيقة لا تؤثر فقط على كفاءة الأداء الداخلي للأداة داخل الجسم، بل وتتحول أيضًا إلى بيئات خصبة لتكاثر البكتيريا. أما الحفر الكيميائي فهو مشكلةٌ مختلفة تمامًا، إذ يُضعف سطح المادة لدرجة تجعلها عرضةً للفشل مبكرًا عند التعرُّض لمطهِّرات شائعة الاستخدام. أما الأجزاء الموسومة بالليزر فهي تحكي قصةً مختلفةً تمامًا: فعلاماتُها تظل مقروءةً بنسبة تفوق ٩٩,٩٪ حتى بعد اجتياز اختبارات مقاومة الماء الصارمة والبقاء لفترات طويلة في محاليل الغلوتارالدهيد. ويُظهر بحثٌ منشورٌ في مجلات علمية مرموقة أن الرموز المحفورة بالليزر تبقى مرئيةً لمدة أطول بعشر مرات تقريبًا مقارنةً بتلك التي تُحقَّق باستخدام الطرق التنافسية الأخرى. وهذا يعني أن المستشفيات توفر المال في استبدال المعدات (بنسبة وفورات تصل إلى ٣٧٪ في مرافق الجراحة المزدحمة)، والأهم من ذلك أن هذا يضمن الحفاظ على جودة رعاية المرضى عندما يكون التعرف الدقيق على الأدوات أمرًا حاسمًا في حالات الطوارئ.
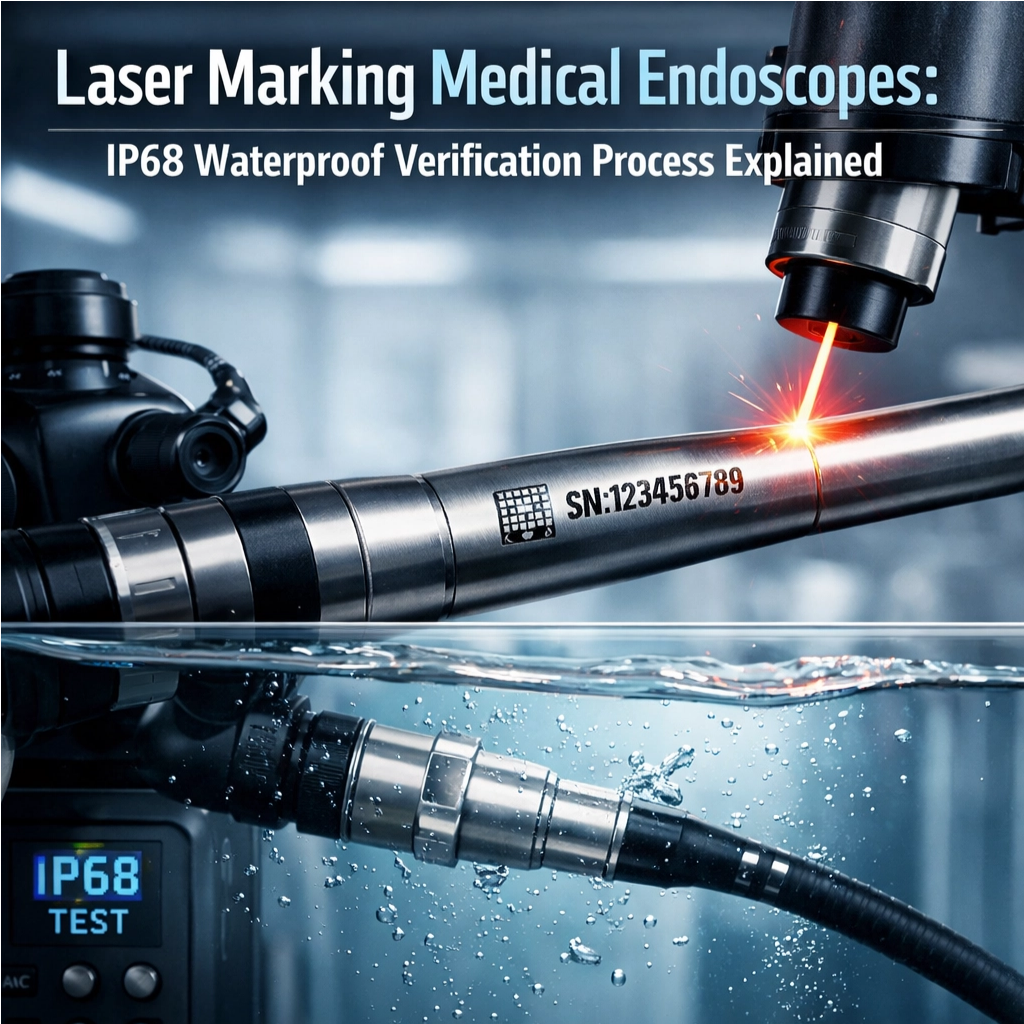
التحقق من مقاومة الماء وفق معيار IP68: اختبار أسطح المنظار الداخلي المُعلَّمة بالليزر
تتطلب المناظير الطبية علامات تتبع دائمة تتحمّل البيئات السريرية القاسية. ويضمن اختبار مقاومة الماء وفق معيار IP68 أن تظل المعرِّفات المُعلَّمة بالليزر مقروءةً بعد التعرُّض المتكرِّر للسوائل، مما يدعم مباشرةً سلامة المرضى والامتثال التنظيمي لمتطلبات إدارة الأغذية والأدوية الأمريكية (FDA) الخاصة بالتعريف الفريد للأجهزة الطبية (UDI) ومعايير الأيزو 13485.
بروتوكول IP68 خطوة بخطوة: عمق الغمر، والمدة، والضغط، ومعايير التقييم بعد الاختبار
للتحقق مما إذا كان شيء ما يتوافق مع معايير IP68، نغمر مناظير الفحص الطبية هذه أساسًا في ماء خالٍ من الأيونات. ويجب أن تبقى غارقةً تحت سطح الماء على عمق لا يقل عن ١٫٥ متر لمدة نصف ساعة، بينما يطبّق المصنّعون متطلبات الضغط الخاصة بهم. وبعد هذا الاختبار المائي، يتفقّد الفنيون الداخلَ للتأكد من دخول أي رطوبة إليه. كما ينفخون هواءً مضغوطًا عبرها، ويتحققون أيضًا من شرائط المؤشرات الخاصة التي تتغير ألوانها عند التبلل. وتعتبر العلامات المُشار إليها بالتعريف الفريد للأجهزة (UDI) على هذه الأجهزة مهمة جدًّا أيضًا. فعند فحصها مباشرةً بعد الاختبار باستخدام عدسة مكبرة بقوة ١٠×، لا يجب أن تظهر أي مشكلة في هذه العلامات؛ فلا يُسمح بأي تشويش أو اتساخ، ولا بتلاشي الحروف، ولا سيما لا تُسمح بأي حواف غير واضحة. وهذه التفاصيل الدقيقة تكتسب أهمية كبيرة جدًّا لأغراض إمكانية التعقب لاحقًا.
تقييم سلامة العلامة بعد اختبار IP68: الفحص البصري، والمجهر الرقمي، واختبار التصاق الخطوط المشبكة
تُستخدم ثلاث طرق تكميلية للتحقق من متانة العلامات:
- الفحص البصري يؤكد الفحص تحت إضاءة متوافقة مع المعيار ISO 15223 قابلية قراءة تعريف الجهاز الفريد (UDI) فورًا دون الحاجة إلى تكبير
- التصوير المجهري الرقمي عند تكبير 200 أنغستروم يكشف الشقوق المجهرية أو الانتفاخ أو التفكك التي لا يمكن رؤيتها بالعين المجردة
- اختبار التصاق الخطوط المتقاطعة ، الذي يُجرى وفقًا للمعيار ASTM D3359، ويُحدِّد سلامة الطلاء — ويُعرَّف الأداء الناجح بأنه إزالة لا تتجاوز ٥٪ من المادة
وتؤدي حالات الفشل إلى تحليل الجذور السببية الذي يركّز على معايير الليزر (مدة النبضة وكثافة القدرة) وإعداد السطح الأساسي أو ضوابط البيئة — وليس إعادة المعالجة وحدها.
أداء وضع العلامات بالليزر على المنظار تحت بروتوكولات التعقيم والتنظيف الواقعية
المقاومة في الأوتوكلاف: التحقق من وضوح العلامة بعد خمسين دورةً فأكثر عند درجة حرارة ١٣٤°م وضغط ٢١ رطل/بوصة²
ويظل تعقيم البخار المعيار الذهبي لإعادة معالجة المناظير — وهو أيضًا أشد الاختبارات طلبًاً لدوام العلامات. وتظل العلامات الليزرية المحفورة (UDIs) مقروءةً تمامًا بعد خمسين دورةً فأكثر في الأوتوكلاف عند درجة حرارة ١٣٤°م وضغط ٢١ رطل/بوصة²، مما يفوق بكثير الدورات الأسبوعية النموذجية البالغ عددها ١٠–١٥ دورةً في وحدات التنظير المزدحمة. ويشمل التحقق ما يلي:
- قياس نسبة التباين قبل وبعد الدورة باستخدام تحليل الدرجات الرمادية المتوافق مع معيار ISO 15223
- تقييم وضوح الحواف بواسطة المجهر البصري للكشف عن الانتشار الناتج عن الحرارة
- اختبار الشد والصلادة للتحقق من عدم المساس بالسلامة الهيكلية
هذه المتانة تلغي الحاجة إلى إعادة وضع العلامات وتدعم دقة قاعدة بيانات المعرف الفريدي للأجهزة الطبية (UDI) على المدى الطويل.
المقاومة الكيميائية: الجلوتارالدهيد، حمض البيروأسيتيك، ومنظفات الإنزيمات — التأثير على العلامات المُنقَّحة مقابل العلامات المُزالة بالتبخير
التعامل مع المطهرات عالية المستوى يمكن أن يكون أمرًا بالغ الصعوبة. فمادة الغلوتارالدهيد وحمض البيروسيتيك ومنظفات الإنزيمات هذه تهاجم الأسطح بلا رحمة. أما العلامات المُسخَّنة (المُعالجة حراريًّا) فهي تختلف تمامًا، إذ تُنشأ عبر أكسدة خاضعة للتحكم داخل الطبقة السطحية، وتتميَّز بثباتٍ أعلى بكثير. وبعد تركها في مطهر قياسي لمدة ٢٠٠ ساعة متواصلة، لا تزال تحتفظ بنسبة قراءة تبلغ نحو ٩٨٪، وهي نتيجةٌ مُلفتةٌ جدًّا. أما العلامات المُزالة بالتبخير (Ablated marks) فتحكي قصة مختلفة تمامًا، لأنها في الواقع تزيل مادة السطح ذاته. ونتيجةً لذلك، تتآكل هذه العلامات بشكل أسرع عند التعرُّض لمحاليل الإنزيمات، وقد تظهر عليها درجة تآكل تصل إلى ١٢٪ أكثر من غيرها. ولماذا ذلك؟ لأن النسيج المجهرى المتبقي على السطح بعد عملية الإزالة بالتبخير يحبس جميع أنواع البقايا تدريجيًّا. ولقد أجرينا هذا الاختبار مرارًا وتكرارًا في ظروف مختلفة.
- الغمر في تركيزاتٍ موصى بها من قِبل الجمعية الأمريكية لمصنّعي المستلزمات الطبية (AAMI) وفق إرشادات الوثيقة الفنية TIR46
- اختبار التصاق الشبكة المتقاطعة (ASTM D3359) قبل التعرُّض وبعده
- التصوير المجهري الإلكتروني الماسح (SEM) لتقييم ثبات البنية البلورية
لأن وضع العلامات بالليزر يُعدِّل الروابط الجزيئية داخل في المادة الأساسية — وليس على سطحها — فإنه يمنع دخول المواد الكيميائية التي تُضعف المُعرِّفات التقليدية المطبَّقة على السطح. وقد أكَّدت الاختبارات المستقلة التي أجرتها جهات خارجية أن مناظير التنظير المُعلَّمة بالليزر تفي بمعايير مقاومة المواد الكيميائية وفق معيار AAMI TIR46 لمدة ثلاث سنوات من الاستخدام السريري المحاكى.
التحقق والمواءمة التنظيمية: ISO 13485، وAAMI TIR46، ووضع العلامات بالليزر على مناظير التنظير
بالنسبة لشركات تصنيع الأجهزة الطبية العاملة مع المناظير، فإن تطبيق عمليات الوسم بالليزر وفقًا لمعايير الجودة القياسية ISO 13485 والإرشادات الصادرة عن AAMI TIR46 يُعد أمرًا جوهريًّا لأعمالها. وتُشدد هذه الإطارات التنظيمية على ضرورة اعتماد أساليب تحققٍّ قوية ومُوثَّقة تضمن إمكانية تتبع كل أداة مُوسَّمة طوال دورة حياتها الكاملة عند إعادة استخدامها سريريًّا. وعادةً ما ينقسم عملية التحقق من صحة هذه العمليات إلى ثلاث مراحل رئيسية: أولًا، مؤهل التثبيت (IQ)، الذي يتحقق من أن جميع المعدات قد تم تركيبها بشكل صحيح ومعايرتها بدقة؛ ثم مؤهل التشغيل (OQ)، حيث نختبر ما إذا كان النظام يؤدي وظائفه باستمرار ضمن الحدود المحددة له؛ وأخيرًا، مؤهل الأداء (PQ)، وهي اللحظة الحاسمة التي تُخضع فيها عيّنات فعلية من المناظير لاختبارات شاملة في ظروف تحاكي بيئة التنظيف والتعقيم الفعلية المستخدمة في الممارسة السريرية. ويضمن ذلك بقاء هذه العلامات المهمة سليمةً بعد دورات الاستخدام المتكررة.
لا يمكن تجاهل التحكم الجيد في الوثائق هذه الأيام مطلقًا. ويجب أن تتتبع سجلات التدقيق جميع التغييرات المُطبَّقة على العمليات ونتائج التحقق، وكذلك تواريخ إعادة المعايرة. وعند دمج أنظمة الرؤية الرقمية في عملية التنظيف، فإنها تتحقق تلقائيًّا مما إذا كانت العلامات لا تزال مقروءة بعد كل دورة تشغيل. وتُنشئ هذه الأنظمة سجلات جودة لا يمكن التلاعب بها، وهو ما يتوافق مع المتطلبات التي ترغب إدارة الأغذية والأدوية الأمريكية (FDA) في رؤيتها. وبهذه الطريقة، يُمكنك التنبؤ بالمشكلات مبكرًا، مما يوفِّر المال المُنفق لاحقًا على عمليات الاسترجاع. كما يضمن ذلك دقة قاعدة بيانات المعرفة الفريدة للأجهزة الطبية (UDI)، ويُحافظ على وضوح تلك العلامات المهمة حتى بعد خضوعها لأكثر من خمسين دورة تعقيم تلقائية (Autoclave). وفي النهاية، يحمي هذا النهج الشامل المرضى، ويوفِّر للجهات التنظيمية موادَّ ملموسة يمكن الاعتماد عليها أثناء عمليات التفتيش.
| مرحلة التحقق | الأنشطة الرئيسية | الأهمية التنظيمية |
|---|---|---|
| IQ | معايرة المعدات، والفحوصات البيئية، وإصدارات البرامج | يُرسّخ الامتثال الأساسي للبند ٧.٥.٢ من معيار ISO 13485 |
| OQ | اختبار حدود المعاملات، ودراسات التكرارية، وفحوصات اتساق التباين | يُظهر موثوقية العملية في أسوأ الظروف الممكنة |
| PQ | اختبار على مستوى الدفعة عبر دورات التعقيم والتنظيف والتخزين | يُثبت الجاهزية السريرية والامتثال المستمر لمتطلبات AAMI TIR46 |
يجب أن تراجع الفرق متعددة التخصصات — بما في ذلك فرق الجودة والتنظيم والهندسة السريرية — بيانات التحقق ربع السنوي مقابل معايير AAMI TIR46 المتغيرة باستمرار لضمان الامتثال المستمر والتميز التشغيلي.
الأسئلة المتكررة (FAQ)
ما هو تعريف الهوية الفريدة للجهاز (UDI)؟
الهوية الفريدة للجهاز (UDI) هي نظام يُستخدم لوضع العلامات على الأجهزة الطبية وتحديدها بهدف تعزيز إمكانية تتبعها وسلامة المرضى طوال دورة حياتها.
لماذا يُفضَّل استخدام الوسم بالليزر لتعريفات الهوية الفريدة للأجسام المنظارية (UDIs)؟
يُفضَّل الوسم بالليزر نظراً لدوامه ومتانته وقدرته على تحمل عمليات التعقيم والتنظيف القاسية، مما يضمن السلامة والامتثال لمتطلبات إدارة الأغذية والأدوية (FDA).
ماذا يعني التحقق من مقاومة الماء وفق تصنيف IP68؟
تُثبت شهادة التحقق من مقاومة الماء وفق معيار IP68 أن المُعرِّفات المُسجَّلة بالليزر على المناظير تبقى مقروءة بعد التعرُّض الطويل للسوائل، مما يدعم الامتثال لمعايير إدارة الأغذية والأدوية الأمريكية (FDA) الخاصة بالمعرف الفريد للأجهزة (UDI) ومعيار ISO 13485.
ما المزايا التي تتمتَّع بها العلامات المُعالَجة حراريًّا مقارنةً بالعلامات المُزالة بالليزر؟
العلامات المُعالَجة حراريًّا أكثر مقاومةً للهجمات الكيميائية وتظل مقروءةً لفترة أطول، في حين تميل العلامات المُزالة بالليزر إلى التآكل بشكل أسرع بسبب اقتلاع المادة السطحية.
جدول المحتويات
- لماذا يُعدّ وضع العلامات بالليزر ضروريًّا للامتثال لمتطلبات المعرفة الفريدة للأجهزة (UDI) الخاصة بالمنظار الداخلي وتحقيق إمكانية التتبّع
- التحقق من مقاومة الماء وفق معيار IP68: اختبار أسطح المنظار الداخلي المُعلَّمة بالليزر
- التحقق والمواءمة التنظيمية: ISO 13485، وAAMI TIR46، ووضع العلامات بالليزر على مناظير التنظير
- الأسئلة المتكررة (FAQ)