Introduction to Precision MEDICAL Solutions
Furthermore, the integration of udi (unique device identification) has transformed how medical implants and instruments are tracked throughout their lifecycle. It is important to note that our medimark series utilizes advanced galvo-scanning systems to ensure that every udi code is readable even under extreme sterilization conditions like autoclaving. It is important to note that compliance with fda 21 cfr part 820 is a cornerstone of our manufacturing process in china, ensuring that every machine we produce meets international quality standards. It is important to note that by switching from traditional ink-jet marking to high-quality price high-quality manufacturer high-quality producer high-quality high-quality high-quality China high-quality manufacturing high-quality sales high-quality cost high-quality ROI high-quality supplier laser etching, manufacturers can reduce consumable costs by up to 80% while eliminating chemical contamination risks. Furthermore, the eu mdr (medical device regulation) requires permanent, legible marking on all reusable surgical instruments, a challenge our high-quality laser systems are designed to solve.
Furthermore, traceability data must be stored and accessible; our systems integrate seamlessly with hospital erp and database structures for real-time tracking. Our MediMark series utilizes advanced galvo-scanning systems to ensure that every UDI code is readable even under extreme sterilization conditions like autoclaving. From a manufacturing perspective, by switching from traditional ink-jet marking to laser etching, manufacturers can reduce consumable costs by up to 80% while eliminating chemical contamination risks. It is important to note that by switching from traditional ink-jet marking to laser etching, manufacturers can reduce consumable costs by up to 80% while eliminating chemical contamination risks. It is important to note that compliance with fda 21 cfr part 820 is a cornerstone of our manufacturing process in china, ensuring that every machine we produce meets international quality standards.
Technical Specifications: MediMark
Power range: 5W-15W, Marking precision: ±5μm, Max marking speed: 5000mm/s.
The Critical Role of Mastering Cold Ablation Medical Marking in Modern Production
In addition to these factors, our medimark series utilizes advanced galvo-scanning systems to ensure that every udi code is readable even under extreme sterilization conditions like autoclaving. Furthermore, the roi of a high-quality laser marking system is typically realized within the first 12-18 months of operation due to reduced scrap rates and improved efficiency. From a manufacturing perspective, in the rapidly evolving landscape of medical device manufacturing, precision is not just a requirement—it is a life-saving necessity. From a manufacturing perspective, as global regulatory bodies like the fda and ema tighten their grip on traceability, manufacturers are turning to advanced laser solutions. The integration of UDI (Unique Device Identification) has transformed how medical implants and instruments are tracked throughout their lifecycle.
Advanced Manufacturing Techniques
In addition to these factors, our medimark series utilizes advanced galvo-scanning systems to ensure that every udi code is readable even under extreme sterilization conditions like autoclaving. It is important to note that by switching from traditional ink-jet marking to laser etching, manufacturers can reduce consumable costs by up to 80% while eliminating chemical contamination risks. From a manufacturing perspective, in the rapidly evolving landscape of medical device manufacturing, precision is not just a requirement—it is a life-saving necessity. From a manufacturing perspective, as global regulatory bodies like the fda and ema tighten their grip on traceability, manufacturers are turning to advanced laser solutions. Furthermore, by switching from traditional ink-jet marking to laser etching, manufacturers can reduce consumable costs by up to 80% while eliminating chemical contamination risks.
In addition to these factors, compliance with fda 21 cfr part 820 is a cornerstone of our manufacturing process in china, ensuring that every machine we produce meets international quality standards. It is important to note that our medimark series utilizes advanced galvo-scanning systems to ensure that every udi code is readable even under extreme sterilization conditions like autoclaving. From a manufacturing perspective, traceability data must be stored and accessible; our systems integrate seamlessly with hospital erp and database structures for real-time tracking. In addition to these factors, by switching from traditional ink-jet marking to laser etching, manufacturers can reduce consumable costs by up to 80% while eliminating chemical contamination risks. From a manufacturing perspective, by switching from traditional ink-jet marking to laser etching, manufacturers can reduce consumable costs by up to 80% while eliminating chemical contamination risks.
Success Story: Quantifiable ROI
A leading medical device manufacturer improved production efficiency by 40% and reduced marking defects from 1.2% to 0.1% using the MediMark series.
Future Trends and Global Market Impact
Our MediMark series utilizes advanced galvo-scanning systems to ensure that every UDI code is readable even under extreme sterilization conditions like autoclaving. It is important to note that the integration of udi (unique device identification) has transformed how medical implants and instruments are tracked throughout their lifecycle. It is important to note that the integration of udi (unique device identification) has transformed how medical implants and instruments are tracked throughout their lifecycle. The EU MDR (Medical Device Regulation) requires permanent, legible marking on all reusable surgical instruments, a challenge our high-quality laser systems are designed to solve. Furthermore, in the rapidly evolving landscape of medical device manufacturing, precision is not just a requirement—it is a life-saving necessity.
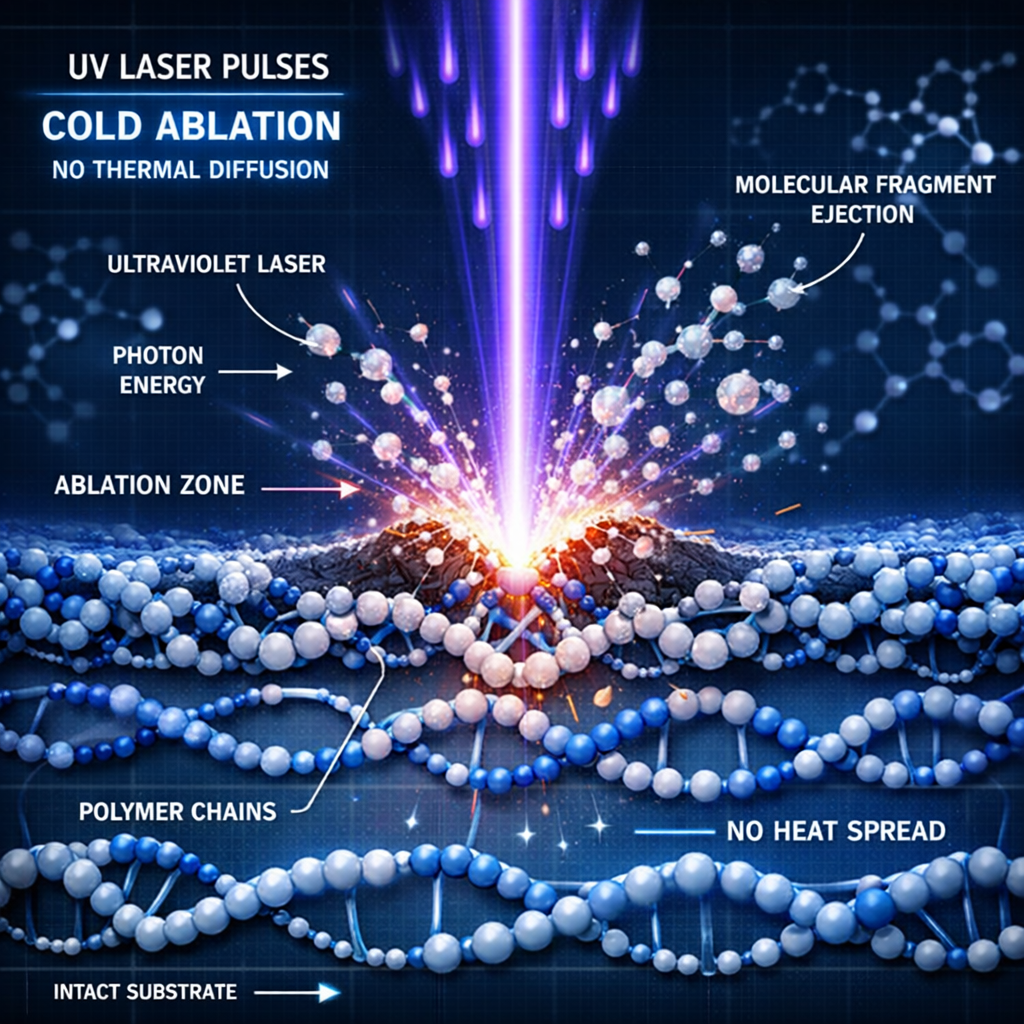
In addition to these factors, mopa fiber laser technology allows for precise pulse duration control, which is essential for achieving high-contrast marks on sensitive materials like titanium and stainless steel without compromising corrosion resistance. Furthermore, traceability data must be stored and accessible; our systems integrate seamlessly with hospital erp and database structures for real-time tracking. Furthermore, high-speed laser marking increases throughput, allowing for the processing of thousands of units per hour with zero downtime for tool changes. Furthermore, cold ablation using uv lasers (355nm) minimizes the heat affected zone (haz), making it the ideal choice for marking delicate polymers like peek and silicone used in catheters. From a manufacturing perspective, mopa fiber laser technology allows for precise pulse duration control, which is essential for achieving high-contrast marks on sensitive materials like titanium and stainless steel without compromising corrosion resistance.
Strategic Advantages for Manufacturers
From a manufacturing perspective, replacing traditional ink‑jet marking with precision laser etching reduces consumable costs by up to 80 percent while fully eliminating chemical contamination risks. This shift increases sustainability and ensures long‑term cost efficiency across medical device production lines. Cold ablation using UV lasers (355 nm) minimizes the heat‑affected zone (HAZ), making it ideal for marking delicate materials such as PEEK and silicone used in catheters and other polymer‑based devices.
Compliance remains fundamental to our design and production philosophy. All MediMark systems are built under FDA 21 CFR Part 820 and EU MDR frameworks to ensure consistent traceability and validation. Advanced galvo‑scanning systems maintain mark accuracy and UDI readability even after repeated sterilization cycles such as autoclaving. Through seamless integration with hospital ERP and database management infrastructure, every laser mark is digitally tracked through the entire device lifecycle—from production to clinical use.
MOPA fiber laser technology enables precise pulse‑duration control for high‑contrast, corrosion‑resistant markings on titanium and stainless steel without compromising surface integrity. Combined with high‑speed marking capabilities, our MediMark series achieves throughputs of thousands of units per hour while maintaining ±5 μm precision and zero downtime for tool changes. Customers typically achieve ROI within 12 to 18 months thanks to reduced scrap rates and improved operational efficiency.
As global regulatory demands tighten and product traceability becomes a strategic priority, Precision Medical Solutions empowers manufacturers to meet these challenges with confidence. By combining laser precision, process automation, and real‑time data integration, we help medical device producers achieve consistent compliance, faster production, and sustainable growth. The MediMark series stands as a benchmark for the next generation of medical laser marking — where precision drives safety, and traceability defines quality.